Volume: 100 ml
Storage: ≤ –15°C
Sterility: Sterile-filtered
Stable Glutamine Solution (L-Alanyl-L-Glutamine, 200 mM) is a highly stable dipeptide form of L-glutamine designed as a direct replacement for conventional L-glutamine in cell culture media. L-glutamine is an essential amino acid and a major energy source for cultured cells, playing a critical role in cell growth, metabolism, and protein synthesis.
Application and Benefits
In standard liquid media, L-glutamine degrades relatively quickly at 37°C, leading to the formation of toxic byproducts such as ammonium ions that can negatively affect cell viability and experimental outcomes. Stable glutamine overcomes this limitation by providing a non-degrading dipeptide form that remains intact in culture conditions.
Cells enzymatically cleave the dipeptide bond to release L-glutamine as needed, ensuring a continuous fresh supply while preventing the accumulation of harmful waste products. This makes the solution particularly advantageous for long-term cell cultures and high-density growth systems.
Formulation and Usage
The L-Alanyl-L-Glutamine solution is prepared in cell culture–grade water at a concentration of 200 mM and is sterile-filtered for contamination-sensitive applications. It can be diluted directly into complete media according to experimental requirements. Store at ≤ –15°C and avoid repeated freeze-thaw cycles to maintain product stability.
For research use only. Not for use in diagnostic or therapeutic procedures. Not for use in humans or animals.
Volume: 100 ml
Storage: +2°C to +8°C
Sterility: Sterile-filtered
HEPES Buffer Solution (1 M), also known as N-2-hydroxyethylpiperazine-N-2-ethanesulfonic acid, is a zwitterionic organic buffering agent widely used in cell culture media. It is designed to maintain stable pH conditions in the physiological range of 6.7 to 8.6, supporting optimal cellular function during in vitro applications.
Application and Benefits
HEPES provides reliable buffering capacity in cell culture systems, particularly when cells are handled outside a CO₂ incubator. The addition of 10–25 mM HEPES to culture media offers enhanced pH stability during extended manipulation periods, helping to maintain consistent experimental conditions.
This buffer is membrane-impermeable, has minimal interference with biochemical reactions, and demonstrates strong chemical and enzymatic stability. These properties make it suitable for a wide range of cell culture and biochemical applications.
Formulation and Usage
The solution is supplied as a 1 M concentrate prepared in cell culture–grade water and is sterile-filtered for use in contamination-sensitive environments. It can be diluted to the desired working concentration depending on application requirements. Store at +2°C to +8°C and handle under aseptic conditions to preserve product integrity.
For research use only. Not for use in diagnostic or therapeutic procedures. Not for use in humans or animals.
Volume: 50 ml
Storage: +2°C to +8°C
Sterility: Sterile-filtered
D-(+)-Glucose Solution (Dextrose Solution) is a sterile, ready-to-use supplement containing the naturally occurring sugar D-(+)-glucose, a central component of cellular metabolism. Glucose is involved in essential biological processes such as energy production, glycosylation, and the formation of glycans that contribute to cellular structure and function.
Application and Benefits
This glucose solution is widely used as a supplement in cell culture media and in numerous cellular and molecular biology applications. As a primary carbon and energy source, glucose supports cell growth, proliferation, and metabolic activity. Its involvement in biosynthetic pathways also makes it critical for maintaining normal cellular physiology and experimental consistency.
Formulation and Usage
The solution is supplied at a high concentration of 250 g/L glucose, allowing flexible dilution into culture media according to experimental requirements. It is sterile-filtered to ensure suitability for contamination-sensitive applications. Store at +2°C to +8°C and handle aseptically to maintain product quality and stability.
For research use only. Not for use in diagnostic or therapeutic procedures. Not for use in humans or animals.
Volume: 10 ml
Storage: +2°C to +8°C
Sterility: Sterile-filtered
Insulin-Transferrin-Selenium (ITS) Solution (100x) is a chemically defined supplement designed for a wide range of cell culture applications. It is most commonly used as an additive to basal cell culture media to support cell growth under reduced-serum or serum-free conditions.
Application and Benefits
Our ITS supplement provides essential components required for the optimal performance of serum-free media. By supplementing conventional nutrient media with ITS, the fetal bovine serum (FBS) requirement for the routine maintenance of many cell lines can be significantly reduced. This helps minimize variability associated with serum use while maintaining consistent cell growth and viability.
Insulin supports cellular uptake and metabolism of key nutrients, transferrin facilitates iron transport, and selenium contributes to antioxidant defense and enzymatic activity. Together, these components promote balanced cell metabolism and improved reproducibility in defined culture systems.
Formulation and Usage
Insulin-Transferrin-Selenium (ITS) is supplied as a 100x concentrated solution in Earle's Balanced Salt Solution (EBSS) without phenol red. For standard applications, dilute 1:100 into the appropriate basal medium to achieve the recommended working concentration. Store at +2°C to +8°C and handle under aseptic conditions to maintain product stability and sterility.
For research use only. Not for use in diagnostic or therapeutic procedures. Not for use in humans or animals.
Volume: 5 ml
Storage: +2°C to +8°C
Sterility: Sterile-filtered
Insulin Human Recombinant Solution is a chemically defined supplement commonly used for the cultivation of mammalian cell lines, including Chinese Hamster Ovary (CHO) cells. This cell culture–grade solution contains recombinant human insulin expressed in Saccharomyces cerevisiae, ensuring high purity and consistent performance in research applications.
Application and Benefits
Insulin is routinely added to serum-free and chemically defined media to promote cell growth and productivity. As a key regulatory hormone, insulin supports cellular uptake, utilization, and storage of glucose, amino acids, and fatty acids. It also inhibits the breakdown of glycogen, proteins, and lipids, thereby contributing to improved cell viability and metabolic stability in culture systems. The chemically defined formulation supports reproducibility and minimizes variability in sensitive cell culture workflows.
Biological Properties and Usage
Insulin is a two-chain polypeptide hormone naturally produced by the β-cells of the pancreatic islets. It has a molecular weight of approximately 5,800 Da. The α
- and β-chains are linked by two interchain disulfide bonds, and the α-chain contains one intrachain disulfide bond. For cell culture applications, the solution should be handled under aseptic conditions and stored at +2°C to +8°C to maintain stability and performance.
For research use only. Not for use in diagnostic or therapeutic procedures. Not for use in humans or animals.
Volume: 100 ml
Storage: +2°C to +8°C
Sterility: Sterile-filtered
Sodium Pyruvate Solution (100 mM) is a sterile, ready-to-use supplement designed to provide an additional, easily accessible energy source for cell culture media. Sodium pyruvate plays a key role in cellular energy metabolism and supports the growth of metabolically active and rapidly proliferating cells, such as tumor cells. Supplementation can enhance cell viability and help maintain metabolic stability in culture systems.
Application and Benefits
This solution is widely used in routine cell culture to enrich media with pyruvate and promote optimal growth conditions. It supports ATP production, may help reduce oxidative stress, and contributes to improved metabolic performance of cultured cells. Manufactured in cell culture–grade water and sterile-filtered, the product ensures consistent quality and reproducibility in research workflows.
Usage and Compatibility
The recommended final concentration for most cell culture applications is 1 mM sodium pyruvate, achieved by diluting the 100 mM stock solution 1:100 into complete culture medium. The solution is compatible with a wide range of basal media and mammalian cell lines. Store at +2°C to +8°C and protect from contamination to maintain product stability.
For research use only. Not for use in diagnostic or therapeutic procedures. Not for use in humans or animals.
Volume: 100 ml
Storage: ≤-15°C
Sterility: Sterile-filtered
Antibiotic/Antimycotic Solution (100x) is a sterile, ready-to-use concentrate designed to reduce microbial contamination risks in cell culture and related laboratory applications. This 100x solution contains a well-established combination of penicillin, streptomycin, and amphotericin B—providing broad-spectrum antimicrobial activity against Gram-positive and Gram-negative bacteria, yeasts, and filamentous fungi. The formulation is suitable for use in eukaryotic cell cultures, bacterial media, and other contamination-sensitive systems, supporting clean and consistent lab operations.
Application and Benefits Optimized for routine research protocols, this solution is widely used to maintain aseptic conditions in cell culture workflows. It offers reliable performance in contamination-sensitive environments, helping researchers reduce the risk of microbial overgrowth without compromising cell health or experimental reproducibility. The sterile-filtered formulation eliminates the need for additional solubilization steps, supporting streamlined media preparation and reducing variability in daily lab procedures.
Usage and Compatibility To achieve standard working concentrations, dilute the solution 1:100 into your complete culture medium. The product is compatible with a broad range of mammalian cell lines and basal media. With consistent stock availability, researchers benefit from dependable supply continuity and simplified logistics planning. The solution should be stored at ≤ –15 °C and protected from repeated freeze-thaw cycles to maintain stability. For research use only. Not for use in diagnostic or therapeutic procedures. Not for use in humans or animals.
Volume: 100 ml
Storage: +2°C to +8°C
Sterility: Sterile-filtered
MEM Non-Essential Amino Acids (100x) is a sterile supplement designed to enhance cell growth and viability in mammalian cell culture systems. The formulation corresponds to a 100x concentrate of the non-essential amino acids found in standard Minimum Essential Medium (MEM), allowing for direct supplementation of basal media with minimal preparation.
Application and Benefits This supplement provides an additional amino acid pool for rapidly proliferating cells or for cell lines that have lost the capacity to synthesize non-essential amino acids de novo. By alleviating the metabolic burden of biosynthesis, it supports improved growth kinetics, prolonged viability, and greater experimental consistency—especially in nutrient-sensitive or high-density cultures.
Composition and Use The solution includes glycine, L-alanine, L-asparagine, L-aspartic acid, L-glutamic acid, L-proline, and L-serine. It is compatible with MEM and most other standard media. To use, dilute 1:100 into the final culture medium. This product is sterile-filtered and ready to use without additional handling steps. For research use only. Not for use in diagnostic or therapeutic procedures. Not for use in humans or animals.
Accutase is a ready-to-use, sterile-filtered cell detachment solution designed as a gentle alternative to trypsin/EDTA for dissociating adherent mammalian cells from standard tissue culture plasticware and adhesion-coated surfaces. It combines proteolytic and collagenolytic enzyme activity in a balanced salt solution to deliver effective yet controlled dissociation, preserving cell-surface proteins and supporting high post-passage viability and rapid reattachment.
The Accutase formulation is based on Dulbecco’s phosphate-buffered saline (DPBS) with EDTA and phenol red as a visual pH indicator. The enzymes are of non-mammalian and non-bacterial origin, making Accutase particularly well suited to stem cell research, vaccine workflows, and any application where animal
- or microbially-derived contaminants must be minimised. The solution auto-inhibits at 37 °C, so no neutralising reagent or serum-containing medium is required after detachment – cells can be transferred directly into fresh medium.
Key Features
Ready-to-use 1x sterile-filtered liquid – no dilution or reconstitution required
Combined proteolytic and collagenolytic enzyme activity for gentle dissociation
Each batch standardised to a defined dissociation activity for lot-to-lot consistency
Non-mammalian and non-bacterial enzyme origin
Auto-inhibits at 37 °C – no neutralising solution needed
Formulated in Dulbecco’s PBS with EDTA
Phenol red included as visual pH indicator
pH 6.8 – 7.8
Typical Applications
Accutase gently dissociates a wide variety of adherent and sensitive cell types, including human embryonic stem cells (hESCs), human induced pluripotent stem cells (iPSCs), neural stem cells, primary neurons, and routinely cultured adherent lines such as HeLa, HEK 293, CHO, MDCK, Vero, NIH/3T3, BHK-21 and A549. Typical use cases include:
Routine subculture and passaging of adherent mammalian cells
Gentle single-cell dissociation of hESCs, iPSCs and other sensitive lines
Sample preparation for flow cytometry and FACS analysis
Analysis of cell-surface markers where epitope integrity matters
Cell migration, proliferation and apoptosis assays
Quiescence assays by serum starvation and oncogene transfection studies
Tumor cell and neural crest cell migration assays
Production scale-up in bioreactor workflows
For routine work, apply approximately 10 ml of Accutase per 75 cm2 of culture surface and incubate for 5–10 minutes at room temperature. The optimal incubation time should be determined for each cell line and should not exceed one hour. Prior to addition, rinse the cell layer with a Ca2+/Mg2+-free salt solution such as DPBS without calcium and magnesium to remove residual serum and divalent cations.
Handling & Storage
Store the unopened bottle frozen at -15 °C or below. Thaw either at room temperature or overnight at +2 °C to +8 °C. Do not thaw Accutase in a 37 °C water bath, as elevated temperatures reduce enzyme activity. After thawing, the solution can be stored for up to 2 months at +2 °C to +8 °C; do not store at room temperature. Do not pre-warm the reagent to 37 °C before application – add it directly to washed cells at room temperature. For long-term shelf life, single-use aliquoting is recommended to avoid repeated thaw cycles. Always work under aseptic conditions.
Quality
Manufactured under strict quality standards. Each batch of Accutase is sterile-filtered and tested for sterility, pH, appearance and dissociation activity to ensure consistent, reproducible performance from lot to lot.
Product Specifications
Specification
Detail
Product typeCell detachment / dissociation reagent
FormatSterile-filtered liquid, ready-to-use
Volume100 ml
Working concentration1x (ready-to-use)
Enzyme activityCombined proteolytic and collagenolytic
Enzyme originNon-mammalian and non-bacterial
Buffer systemDulbecco’s PBS with EDTA
pH indicatorPhenol red
pH range6.8 – 7.8
AppearanceClear, pale red to orange solution
Storage temperature-15 °C or below
Stability after thawingUp to 2 months at +2 °C to +8 °C
Recommended use volume~10 ml per 75 cm2 culture surface
Typical incubation time5 – 10 minutes at room temperature
Shipping conditionsFrozen on dry ice
Intended useFor research use and further manufacturing only
Formulation (Composition per Liter)
Component
Concentration (mg/L)
Inorganic Salts
Sodium chloride (NaCl)8000.00
Disodium hydrogen phosphate (Na2HPO4)1150.00
Potassium chloride (KCl)200.00
Potassium dihydrogen phosphate (KH2PO4)200.00
Other Components
EDTA · 4Na (tetrasodium EDTA)220.00
Phenol red3.00
Proprietary enzyme blend (proteolytic and collagenolytic activity)1x
Accutase is a registered trademark of Innovative Cell Technologies, Inc.
This ready-to-use, sterile-filtered liquid formulation is supplemented with Earle’s Balanced Salt Solution (EBSS), 2 mM L-glutamine, D-glucose (1.0 g/L), and 2.2 g/L sodium bicarbonate (NaHCO3), making it suitable for use in a CO2-controlled incubator atmosphere (typically 5 % CO2). The included phenol red acts as a pH indicator, allowing convenient visual monitoring of medium condition during cell culture.
Key Features
Classic Eagle’s MEM formulation with Earle’s Balanced Salt Solution (EBSS)
2 mM L-glutamine included – ready for immediate use
2.2 g/L sodium bicarbonate – buffered for 5 % CO2 incubation
With D-glucose (1.0 g/L) as primary carbon source
With phenol red as pH indicator
Without HEPES and without sodium pyruvate
Sterile-filtered liquid medium, ready to use
pH 7.0 – 7.6
Typical Applications
EMEM supports the cultivation of a wide variety of mammalian cell lines, including HeLa, HEK 293, Vero, MRC-5, L-929, BHK-21, and many primary cells. Common applications include:
Routine maintenance and expansion of adherent cell lines
Virus propagation and vaccine production workflows
Cytotoxicity and bioassay applications
Transfection and protein expression studies
Foundational research in cell biology and molecular biology
For optimal cell growth, EMEM is typically supplemented with 5–10 % fetal bovine serum (FBS) and, depending on the cell line, with non-essential amino acids (NEAA) and antibiotics such as penicillin/streptomycin.
Handling & Storage
Store the unopened bottle at +2 °C to +8 °C, protected from light. After opening, use under aseptic conditions. L-Glutamine in solution is subject to gradual degradation – we recommend using the medium within 4 weeks of opening for best performance, or supplementing with fresh L-glutamine prior to use if stored for longer periods. Allow the medium to warm to 37 °C before adding it to cells.
Quality
Manufactured under strict quality standards. Each batch is tested for sterility, pH, osmolality, and endotoxin levels to ensure consistent performance in cell culture applications.
Product Specifications
Specification
Detail
Product typeMEM
Product categoryCell culture media
FormatLiquid
SterileYes
Size500 ml
L-GlutamineWith L-glutamine (2 mM)
GlucoseWith glucose (1.0 g/L)
Sodium bicarbonateWith NaHCO3 (2.2 g/L)
HEPESWithout HEPES
Sodium pyruvateWithout sodium pyruvate
Phenol redWith phenol red
Salt solutionEarle’s Balanced Salt Solution (EBSS)
pH7.0 – 7.6
Endotoxin contentNot specified
Storage+2 °C to +8 °C
Formulation (Composition per Liter)
Component
Concentration (mg/L)
Inorganic Salts
Calcium chloride · 2H2O265.00
Magnesium sulfate97.72
Potassium chloride400.00
Sodium chloride6,800.00
Sodium dihydrogen phosphate, anhydrous122.00
Sodium bicarbonate (NaHCO3)2,200.00
Amino Acids
L-Arginine · HCl126.00
L-Cystine · 2HCl31.30
L-Glutamine292.00
L-Histidine · HCl · H2O42.00
L-Isoleucine52.00
L-Leucine52.00
L-Lysine · HCl72.50
L-Methionine15.00
L-Phenylalanine32.00
L-Threonine48.00
L-Tryptophan10.00
L-Tyrosine · 2Na · 2H2O51.90
L-Valine46.00
Vitamins
D-Calcium pantothenate1.00
Choline chloride1.00
Folic acid1.00
myo-Inositol2.00
Nicotinamide1.00
Pyridoxal · HCl1.00
Riboflavin0.10
Thiamine · HCl1.00
Other Components
D(+)-Glucose1,000.00
Phenol red10.00
Key features of Freeze Medium CM-1 include:
Broad Compatibility: Effective for a wide range of cell types, including primary cells, stem cells, and established cell lines.
High Viability: Optimized to maximize post-thaw cell recovery and viability, ensuring reliable experimental outcomes.
Ready-to-Use: Conveniently prepared and sterilized for immediate application, reducing preparation time and risk of contamination.
Enhanced Stability: Maintains consistent performance under standard cryopreservation conditions, ensuring reproducible results.
Long Shelf Life: CM-1 is a serum-containing, ready-to-use cryopreservation medium that can be stored in the refrigerator for up to one year.
Using CM-1 for Freezing Cells
To use CM-1 for freezing both adherent and suspension cells, follow these steps:
For adherent cells, wash and dissociate them from the culture substrate. For suspension cells, proceed directly to the next step.
Count the cells to ensure they are at the proper concentration.
Centrifuge the cells to pellet them, then resuspend in CM-1 freeze medium.
Transfer the resuspended cells into cryovials.
Use a slow-freezing method before transferring the cells to long-term storage.
Method
Description
Steps
❄️
Manual Freezing
A step-by-step method involving gradual temperature reduction to ensure cell viability.
1️⃣ Place cells in freeze medium in a 4°C freezer for 40 minutes.
2️⃣ Transfer to a -80°C freezer for 24 hours.
3️⃣ Store cells in liquid nitrogen for long-term preservation.
❄️
Using Mr. Frosty
A convenient device that allows for controlled freezing rates without electrical power.
1️⃣ Prepare cells in cryovials with freeze medium.
2️⃣ Place cryovials in Mr. Frosty container.
3️⃣ Store at -80°C for 24 hours before transferring to liquid nitrogen.
❄️
Controlled-Rate Freezer
A high-precision freezer by Thermo Fisher or other manufacturers designed for controlled temperature reduction.
1️⃣ Program the device to gradually decrease the temperature.
2️⃣ Place prepared cells in the freezer.
3️⃣ After the freezing cycle, transfer cells to liquid nitrogen.
Store the cryovials at temperatures below -130°C or in liquid nitrogen for long-term preservation.
Ingredients
Contains FBS, DMSO, Glucose, Salts
Buffering capacity: pH = 7.2 to 7.6
Cytion’s Freeze Medium CM-1 offers a reliable solution for cryopreservation, ensuring high cell viability and functionality post-thaw for a wide range of research applications.
Ham's F-12K (Kaighn's) Medium is carefully formulated to optimize cell culture conditions. It features an enriched composition, providing elevated levels of essential components such as amino acids and sodium pyruvate, as well as additional elements including putrescine, thymidine, hypoxanthine, and zinc. These additions enable researchers to supplement the medium with minimal serum or defined components for specific cell types, facilitating precise experimental conditions.
Notably, Ham's F-12K (Kaighn's) Medium does not contain proteins or growth factors. Consequently, supplementation with growth factors and Fetal Bovine Serum (FBS) is often necessary, allowing researchers to tailor the medium to the requirements of their specific cell lines. For optimal performance, the concentration of FBS must be carefully optimized for each cell line, ensuring optimal growth and functionality.
To maintain physiological pH, Ham's F-12K (Kaighn's) Medium employs a sodium bicarbonate buffer system (2.5 g/L), necessitating a controlled 5-10% CO2 environment during cultivation. This ensures the medium's pH remains within the ideal range for cell growth and viability.
Quality control
pH = 7.2 +/
- 0.02 at 20-25°C.
Each lot has been tested for sterility and absence of mycoplasma and bacteria.
Maintenance
Keep refrigerated at +2°C to +8°C in the dark. Freezing as well as warming up to +37°C minimize the quality of the product.
Do not heat the medium to more than 37°C or use uncontrollable sources of heat (e.g., microwave appliances).
If only a part of the medium is to be used, remove this amount from the bottle and warm it up at room temperature.
Shelf life for any medium but the basic medium is 8 weeks from the date of manufacture.
Composition
Components
mg/L
Inorganic Salts
Calcium chloride x 2H2O
135,24
Copper(II) sulfate x 5H2O
0,00
Iron (II) sulfate x 7H2O
0,83
Magnesium chloride x 6H2O
105,72
Magnesium sulfate x 7H2O
394,49
Potassium chloride
283,29
Potassium dihydrogen phosphate
58,52
Sodium chloride
7597,20
di-Sodium hydrogen phosphateanhydrous
115,02
Zinc sulfate x 7H2O
0,14
Other Components
D(+)-Glucose anhydrous
1260,00
Hypoxanthine
4,08
DL-α-Lipoic acid
0,21
Phenol red
3,00
Putrescine x 2HCl
0,32
Sodium pyruvate
220,00
NaHCO3
2500,00
Thymidine
0,73
Amino Acids
L-Alanine
17,82
L-Arginine x HCl
421,40
L-Asparagine x H2O
30,02
L-Aspartic acid
26,62
L-Cysteine x HCl x H2O
70,24
L-Glutamine
292,20
L-Glutamic acid
29,42
Glycine
15,01
L-Histidine x HCl x H2O
41,92
L-Isoleucine
7,87
L-Leucine
26,24
L-Lysine x HCl
73,04
L-Methionine
8,95
L-Phenylalanine
9,91
L-Proline
69,06
L-Serine
21,02
L-Threonine
23,82
L-Tryptophan
4,08
L-Tyrosine
10,87
L-Valine
23,42
Vitamins
D(+)-Biotin
0,07
D-Calcium pantothenate
0,48
Choline chloride
13,96
Folic acid
1,32
myo-Inositol
18,02
Nicotinamide
0,04
Pyridoxine x HCl
0,06
Riboflavin
0,04
Thiamine x HCl
0,34
Vitamin B12
1,36
Phosphate-buffered saline (PBS) is a widely used buffer solution in biological and chemical research. It plays a crucial role in maintaining the pH balance and osmolarity during various experimental procedures, including tissue processing and cell culture. Our PBS solution is meticulously formulated with high-purity ingredients to ensure stability and reliability in every experiment. The osmolarity and ion concentrations of our PBS closely mimic those of the human body, making it isotonic and non-toxic to most cells.
Composition of Our PBS Solution
Our PBS solution is a pH-adjusted blend of ultrapure-grade phosphate buffers and saline solutions. At a 1X working concentration, it contains:
8000 mg/L Sodium chloride (NaCl)
200 mg/L Potassium chloride (KCl)
1150 mg/L Sodium phosphate dibasic anhydrous (Na2HPO4)
200 mg/L Potassium phosphate monobasic anhydrous (KH2PO4)
This composition ensures an optimal pH and ionic balance, suitable for a wide range of biological applications.
Applications of Our PBS Solution
Our PBS solution is ideal for various applications in biological research. Its isotonic and non-toxic properties make it suitable for substance dilution and cell container rinsing. PBS solutions containing EDTA are effective for disengaging attached and clumped cells. However, divalent metals such as zinc should not be added to PBS, as this can cause precipitation. In such cases, Good's buffers are recommended. Additionally, our PBS solution is an acceptable alternative to viral transport medium for the transport and storage of RNA viruses, including SARS-CoV-2.
Quality Control
Sterile-filtered
Storage and Shelf Life
Store at +2°C to +25°C, protected from light.
Once opened, store at 2°C to 25°C and use within 24 months.
Shipping Conditions
Ambient temperature
Maintenance
Keep refrigerated at +2°C to +8°C in the dark. Avoid freezing and frequent warming to +37°C, as it reduces product quality.
Do not heat the medium beyond 37°C or use uncontrolled heat sources such as microwave appliances.
If only part of the medium is to be used, remove the required amount and warm it to room temperature before use.
Composition
Category
Components
Concentration (mg/L)
Salts
Potassium chloride
200
Potassium phosphate monobasic anhydrous
200
Sodium chloride
8000
Sodium phosphate dibasic anhydrous
1150
Initially designed to support the growth of human leukemic cells in both suspension and monolayer cultures, RPMI 1640 Medium has evolved through modifications by researchers and commercial suppliers to become suitable for a diverse range of mammalian cells. It is exceptionally compatible with cell lines such as HeLa, Jurkat, MCF-7, PC12, PBMC, astrocytes, and carcinomas.
RPMI 1640 Medium stands apart from other cell culture media due to its unique composition. It contains a substantial amount of phosphate, amino acids, and vitamins. Notably, it encompasses biotin, vitamin B12, and PABA, absent in Eagle's Minimal Essential Medium or Dulbecco's Modified Eagle Medium. Moreover, RPMI 1640 Medium exhibits significantly elevated concentrations of vitamins inositol and choline. However, it does not contain proteins, lipids, or growth factors. Consequently, supplementation with 10% Fetal Bovine Serum (FBS) is commonly required to provide optimal conditions for cell growth.
The buffering system of RPMI 1640 Medium relies on sodium bicarbonate and necessitates a 5-10% CO2 environment to maintain a physiologically appropriate pH. The inclusion of the reducing agent glutathione further distinguishes this medium from others.
Quality Control
Sterile-filtered
Storage and Shelf Life
Store at +2°C to +8°C, protected from light.
Once opened, store at 4°C and use within 6–8 weeks.
Shipping Conditions
Ambient temperature
Maintenance
Keep refrigerated at +2°C to +8°C in the dark. Avoid freezing and frequent warming to +37°C, as it reduces product quality.
Do not heat the medium beyond 37°C or use uncontrolled heat sources such as microwave appliances.
If only part of the medium is to be used, remove the required amount and warm it to room temperature before use.
Composition
Category
Components
Concentration (mg/L)
Amino Acids
Glycine
10.00
L-Alanyl-L-Glutamine
434.40
L-Arginine
200.00
L-Asparagine H2O
56.82
L-Aspartic Acid
20.00
L-Cystine 2HCl
65.20
L-Glutamic Acid
20.00
L-Histidine HCl H2O
20.27
L-Hydroxy-L-Proline
20.00
L-Isoleucine
50.00
L-Leucine
50.00
L-Lysine HCl
40.00
L-Methionine
15.00
L-Phenylalanine
15.00
L-Proline
20.00
L-Serine
30.00
L-Threonine
20.00
L-Tryptophan
5.00
L-Tyrosine 2Na 2H2O
28.83
L-Valine
20.00
Vitamins
p-Amino Benzoic Acid
1.00
D-Biotin
0.20
Choline Chloride
3.00
D-Calcium Pantothenate
0.25
Folic Acid
1.00
myo-Inositol
35.00
Nicotinamide
1.00
Pyridoxine HCl
1.00
Riboflavin
0.20
Thiamine HCl
1.00
Vitamin B12
0.005
Inorganic Salts
Ca(NO3)2 4H2O
100.00
KCl
400.00
MgSO4 7H2O
100.00
NaCl
6000.00
NaHCO3
2000.00
Na2HPO4
800.00
Other Components
D-Glucose
2000.00
L-Glutathione Reduced
1.00
Phenol Red Sodium Salt
5.30
This unique formulation combines Dulbecco's Modified Eagle Medium (DMEM) and Ham's F-12 (Ham's Nutrient Mixture F-12) in a precise 1:1 ratio. The addition of L-glutamine further enhances its composition.
DMEM, derived from Eagle's Minimal Essential Medium (EMEM), offers an increased concentration of amino acids and vitamins compared to its predecessor. In contrast, Ham's F-12 is based on Ham's F-10 medium, providing a complementary set of essential components.
To support optimal cell growth, it is common practice to supplement DMEM:Ham's F12 with FBS at a typical concentration of 5-10%. This addition is necessary as the medium lacks growth hormones, lipids, and proteins crucial for cellular development.
DMEM:Ham's F12 incorporates a pH buffer system and is often supplemented with phenol red, a pH indicator. Cultured cells in DMEM:Ham's F12, or any medium utilizing the bicarbonate buffer system, require a controlled CO2 environment of 5-10% to maintain appropriate pH levels.
Quality Control
Sterile-filtered
Storage and Shelf Life
Store at +2°C to +8°C, protected from light.
Once opened, store at 4°C and use within 6–8 weeks.
Shipping Conditions
Ambient temperature
Maintenance
Keep refrigerated at +2°C to +8°C in the dark. Avoid freezing and frequent warming to +37°C, as it reduces product quality.
Do not heat the medium beyond 37°C or use uncontrolled heat sources such as microwave appliances.
If only part of the medium is to be used, remove the required amount and warm it to room temperature before use.
Composition
Category
Components
Concentration (mg/L)
Amino Acids
Glycine
18.75
L-Alanine
4.45
L-Arginine HCl
147.50
L-Asparagine H₂O
7.50
L-Aspartic Acid
6.65
L-Cysteine HCl H₂O
17.56
L-Cystine 2 HCl
31.29
L-Glutamic Acid
7.35
L-Glutamine
365.00
L-Histidine HCl H₂O
31.48
L-Isoleucine
54.47
L-Leucine
59.05
L-Lysine HCl
91.25
L-Methionine
17.24
L-Phenylalanine
35.48
L-Proline
17.25
L-Serine
26.25
L-Threonine
53.45
L-Tryptophan
9.02
L-Tyrosine 2 Na 2 H2O
55.79
L-Valine
52.85
Vitamins
D-Biotin
0.0035
Choline Chloride
8.98
D-Calcium Pantothenate
2.24
Folic Acid
2.66
myo-Inositol
12.60
Nicotinamide
2.02
Pyridoxine HCl
0.031
Pyridoxal HCl
2.00
Riboflavin
0.219
Thiamine HCl
2.17
Vitamin B12
0.68
Inorganic Salts
CaCl2 2 H2O
154.50
CuSO4 5 H2O
0.0013
Fe(NO3)3 9 H2O
0.05
FeSO4 7 H2O
0.417
KCl
311.80
MgCl2 6 H2O
61.20
MgSO4 7 H2O
100.00
NaCl
6996.00
NaHCO3
1200.00
Na2HPO4
71.02
NaH2PO4 2 H2O
70.87
ZnSO4 7 H2O
0.432
Other Components
D-Glucose
3151.00
Hypoxanthine
2.40
HEPES
3574.50
Linoleic Acid
0.042
Lipoic Acid
0.105
Phenol Red Sodium Salt
8.63
Putrescine 2 HCl
0.081
Sodium Pyruvate
55.00
Thymidine
0.365
- 0.02 at 20-25°C. Each lot has been tested for sterility and absence of mycoplasma and bacteria. Maintenance Keep refrigerated at +2°C to +8°C in the dark. Freezing and warming up to +37°C minimize the quality of the product. Do not heat the medium to more than 37°C or use uncontrollable sources of heat (e.g., microwave appliances). If only a part of the medium is to be used, remove this amount from the bottle and warm it up at room temperature. Shelf life for any medium except for the basic medium is 6 to 8 weeks from the date of opening. Composition Components mg/L Inorganic SaltsCalcium chloride x 2H2O132.00 Magnesium sulfate97.67 Potassium chloride400.00 Sodium chloride6,460.00 Disodium hydrogen phosphate (anhydrous)504.00 Other ComponentsD(+)-Glucose (anhydrous)3,000.00 Glutathione (reduced)0.50 Meat Peptone600.00 Phenol red sodium salt11.00 Amino AcidsL-Alanine13.36 L-Arginine x HCl42.14 L-Asparagine x H2O45.03 L-Aspartic acid19.97 L-Cysteine x HCl x H2O31.75 L-Glutamine (stable)219.15 L-Glutamic acid22.07 Glycine7.51 L-Histidine x HCl x H2O20.96 L-Hydroxyproline19.67 L-Isoleucine39.36 L-Leucine39.36 L-Lysine x HCl36.54 L-Methionine14.92 L-Phenylalanine16.52 L-Proline17.27 L-Serine26.28 L-Threonine17.87 L-Tryptophan3.06 L-Tyrosine disodium salt26.10 L-Valine17.57 Vitaminsp-Aminobenzoic acid1.00 Ascorbic acid0.56 D(+)-Biotin0.20 D-Calcium pantothenate0.20 Choline chloride5.00 Folic acid10.00 myo-Inositol36.00 Nicotinamide0.50 Nicotinic acid0.50 Pyridoxal HCl0.50 Pyridoxine HCl0.50 Riboflavin0.20 Thiamine HCl0.20 Vitamin B122.00
Medium 199 offers a range of applications in the field. It can effectively maintain the cumulus-oocyte complex (COC) and support the in vitro maturation of oocytes. Additionally, it is employed in the rinsing of aspiration lines during ovum collection from German Holstein cows. Moreover, Medium 199 serves as an excellent medium for the culture of cardiac endothelial cells derived from rats. These applications demonstrate the versatility and adaptability of Medium 199 to various experimental needs.
History
The development of Medium 199 in the 1950s marked a significant advancement in tissue culture media. Prior to its introduction, many culture media relied on animal-derived products and tissue extracts. However, Morgan and colleagues revolutionized the field by formulating a completely defined nutritional source for cell cultures. Through their experiments involving different combinations of vitamins, amino acids, and other factors, they discovered the exceptional growth-promoting properties of Medium 199.
Quality control
pH = 7.2 +/
- 0.02 at 20-25°C.
Each lot has been tested for sterility and absence of mycoplasma and bacteria.
Maintenance
Keep refrigerated at +2°C to +8°C in the dark. Freezing and warming up to +37° C minimize the quality of the product.
Do not heat the medium to more than 37° C or use uncontrollable sources of heat (e.g., microwave appliances).
If only a part of the medium is to be used, remove this amount from the bottle and warm it up at room temperature.
Shelf life for any medium except for the basic medium is 8 weeks from the date of manufacture.
Composition
Components
mg/L
Inorganic Salts
Calcium chloride x 2H2O
264,92
Iron (III) nitrate x 9H2O
0,72
Magnesium sulfate
97,67
Potassium chloride
400,00
Sodium acetat x 3H2O
82,95
Sodium chloride
6,800.00
Sodium dihydrogen phosphate x H2O
140,00
Other Components
Adenine sulfate
10,00
AMP
0,20
ATP
1,00
Cholesterol
0,20
2‘-Deoxyribose
0,50
D(+)-Glucose anhydrous
1,000.00
Glutathione (red.)
0,05
Guanine x HCl
0,30
Hypoxanthine
0,30
Phenol red
10,00
D-Ribose
0,50
Thymine
0,30
Tween 80
4,90
Uracil
0,30
Xanthine
0,30
NaHCO3
2,200.00
Amino Acids
L-Alanine
25,00
L-Arginine x HCl
70,00
L-Aspartic acid
30,00
L-Cysteine x HCl x H2O
0,10
L-Cystine
20,00
L-Glutamine stable
149,00
L-Glutamic acid
67,00
Glycine
50,00
L-Histidine x HCl x H2O
21,88
L-Hydroxyproline
10,00
L-Isoleucine
20,00
L-Leucine
60,00
L-Lysine x HCl
70,00
L-Methionine
15,00
L-Phenylalanine
25,00
L-Proline
40,00
L-Serine
25,00
L-Threonine
30,00
L-Tryptophan
10,00
L-Tyrosine
40,00
L-Valine
25,00
Vitamins
4-Amino benzoic acid
0,05
Ascorbic acid
0,05
D(+)-Biotin
0,01
Calciferol
0,10
D-Calcium pantothenate
0,01
Choline chloride
0,50
Folic acid
0,01
myo-Inositol
0,05
Menadione
0,01
Nicotinic acid
0.025
Nicotinamide
0.025
Pyridoxal x HCl
0.025
Pyridoxol x HCl
0.025
Riboflavin
0,01
DL-α-Tocopherol phosphate disodium salt
0,01
Thiamine x HCl
0,01
Vitamine A acetate
0,14
IMDM is well suited for rapidly proliferating, high-density cell cultures, including Jurkat, COS-7, and macrophage cells. The various modifications of IMDM available for a range of cell culture applications can be found using the media selector tool. Liquid media provide essential nutrients for all cell culture applications. Each of our high-quality cell culture media is manufactured according to the initially published formula or modifications necessary to the consistent performance and stability of the culture medium.
IMDM vs. DMEM
IMDM contains potassium nitrate instead of ferric nitrate and HEPES and sodium pyruvate. The additional components in IMDM make it more suitable for specialized cell types and specific applications than DMEM.
IMDM vs. RPMI
IMDM and RPMI have different formulations that may be relevant for PMA/ionomycin stimulation. One significant difference is the concentration of Ca2+. While RPMI contains 0.42 mM Ca2+, IMDM contains 1.49 mM.
Quality control
pH = 7.2 +/
- 0.02 at 20-25°C.
Each lot has been tested for sterility and absence of mycoplasma and bacteria.
Maintenance
Keep refrigerated at +2°C to +8°C in the dark. Freezing and warming up to +37° C minimize the quality of the product.
Do not heat the medium to more than 37° C or use uncontrollable sources of heat (e.g., microwave appliances).
If only a part of the medium is to be used, remove this amount from the bottle and warm it up at room temperature.
Shelf life for any medium except for the basic medium is 8 weeks from the date of manufacture.
Composition
Components
mg/L
Inorganic Salts
Calcium chloride x 2 H2O
219,00
Potassium chloride
330,00
Potassium nitrate
0.076
Magnesium sulfate anhydrous
97,73
Sodium chloride
4,505.00
Sodium dihydrogen phosphate anhydrous
109,00
Sodium selenite
0,02
Other Components
D(+)-Glucose anhydrous
4,500.00
HEPES
5,958.00
Sodium pyruvate
110,00
Phenol red
15,00
Amino Acids
L-Alanine
25,00
L-Arginine x HCl
84,00
L-Asparagine x H2O
25,00
L-Aspartic acid
30,00
L-Cystine x 2HCl
91,24
L-Glutamine
584,00
L-Glutamic acid
75,00
Glycine
30,00
L-Histidine x HCl x H2O
42,00
L-Isoleucine
104,80
L-Leucine
104,80
L-Lysine x HCl
146,20
L-Methionine
30,00
L-Phenylalanine
66,00
L-Proline
40,00
L-Serine
42,00
L-Threonine
95,20
L-Tryptophan
16,00
L-Tyrosine x 2Na
104,20
L-Valine
93,60
Vitamins
D(+)-Biotin
0.013
D-Calcium pantothenate
4,00
Choline chloride
4,00
Folic acid
4,00
myo-Inositol
7,20
Cell culture media: An overview
In the field of life sciences, one of the most important methodologies is cell culture. The removal of cells, tissues, or organs from an animal or plant and the subsequent implantation of those cells, tissues, or organs in an artificial environment that is favorable to their survival and/or growth is what is meant by the phrase "cell culture." The fundamental environmental needs for optimal cell development are controlled temperature, a substrate for cell attachment, an adequate growth medium, and an incubator that maintains the optimum pH and osmolality. Cells must have these conditions in order to grow to their full potential.
The selection of an adequate growth medium for in vitro cultivation is the stage in cell culture that is both the most critical and the most vital. A growth medium, also known as culture media, is a liquid or gel formulated to encourage organisms' development on a microscopic, cellular, or plant-like scale. The medium used for cultivating cells often contains an adequate supply of energy and substances that control the cell cycle. The main components of a culture media include amino acids, vitamins, inorganic salts, glucose, and serum. The serum is added to the medium because it acts as a source of growth factors, hormones, and attachment factors. In addition to providing nutrients, the medium also contributes to the upkeep of pH and osmolality levels.
Types of medium used in cell culture
Both human and animal cells have the potential to be grown in either an artificial or synthetic medium or an entirely natural media that is supplemented with natural elements. In the following, we will give you an overview of the different currently available media types.
Natural media
Only biological fluids that exist in their natural state may be found in natural media. Natural media are very helpful and easy for the cultivation of a broad variety of animal cell types. The lack of understanding of the precise components that make up natural media is the primary factor contributing to the low repeatability of results obtained using natural media.
Artificial media
The preparation of artificial or synthetic media involves the addition of nutrients (both organic and inorganic), serum proteins, carbohydrates, cofactors, vitamins, and salts, as well as O2 and CO2 gas phases [1].
Various types of artificial media have been developed in order to fulfill one or more of the following functions: 1) Immediate survival (a balanced salt solution with a precise pH and osmotic pressure). 2) Prolonged survival (a balanced salt solution supplemented with different formulations of organic chemicals and/or serum). 3) Indefinite development. 4) Specialized functions.
There are four distinct classifications for artificial media:
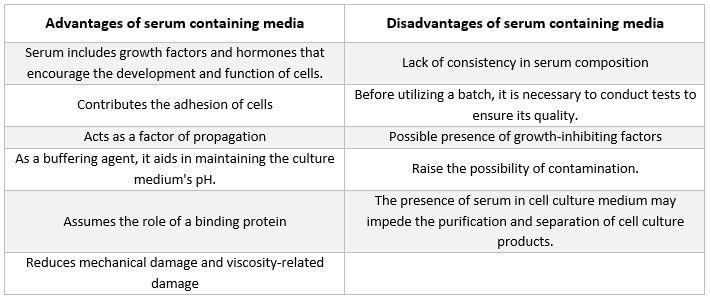
Serum containing media
The most frequent kind of supplement found in medium used for growing animal cells is fetal bovine serum. It is added to the culture medium as a low-cost supplement in order to achieve the best possible growth conditions. In addition to acting as a transporter or chelator for nutrients that are unstable or water-insoluble, hormones and growth factors, protease inhibitors, and other substances, the serum also binds and neutralizes harmful molecules.
Serum-free Medium
The presence of serum in the media has a number of disadvantages and has the potential to cause major errors in interpretation in immunological research [2, 3]. There have been a variety of different serum-free mediums created [4, 5]. These media are generally specifically formulated to support the culture of a single cell type, such as Knockout Serum Replacement and Knockout DMEM from Thermo Fisher Scientific, and mTESR medium from Stem Cell Technologies [6], for stem cells [7].
Additionally, these media incorporate defined quantities of purified growth factors, lipoproteins, and other proteins, which are otherwise typically provided by the serum [8]. These media are often referred to as "defined cultural media" since the components that make up these media are well understood.
Chemically defined media
These media include ultra-pure inorganic and organic components that have not been contaminated by any kind of contamination. They may also include pure protein additions, such as growth factors.
The genetic modification of bacteria or yeast, together with the addition of particular fatty acids, vitamins, cholesterol, and amino acids, results in the production of their component parts [9].
Protein-free media
Protein-free media are those that do not include any protein at all and instead only include non-protein elements. When compared to media with added serum, the use of media without added protein promotes greater cell proliferation and protein expression and makes it easier to purify any product generated in a downstream process [10-12]. Protein is not included in formulations such as MEM and RPMI-1640. However, a protein supplement might be administered if it is necessary.
Culture media and its basic components
Commercial culture medium may be purchased as a powder or a liquid and often include a variety of nutrients such as amino acids, glucose, salts, vitamins, and other dietary supplements.
The needs for these components are different for each cell line, and these variations are responsible for the wide number of different formulations of media. Each component is responsible for a certain function, which will be outlined in the following paragraphs:
Buffering systems
To maintain optimal growing conditions, pH must be controlled, which is often done by one of two buffering systems:
Natural buffering system
The CO2/H2CO3 ratio in the atmosphere is equal to that of the medium, creating a natural buffering mechanism. In order to preserve their natural buffering mechanism, cultures must be kept in an air environment with 5-10% CO2, which is often achieved by using a CO2 incubator. One of the best things about using a natural buffer is how cheap and safe it is.
HEPES
Chemical buffering using the zwitterion HEPES has a greater buffering capability in the pH range of 7.2-7.4 and does not need a regulated gaseous environment. For particular cell types, a larger dose of HEPES may be harmful. Media containing HEPES are likewise much more susceptible to the phototoxic effects of fluorescent light [13].
Phenol Red
The pH indicator phenol red is often included in commercially available culture medium, allowing for continuous monitoring of pH. By expanding the cells, the metabolites produced by these cells cause a shift in pH and therefore a color change of the media. Phenol red has a dual effect on a medium's color, turning it yellow at acidic pH and purple at alkaline pH. pH 7.4, the optimal value for cell culture, causes the medium to appear fluorescent red.
But phenol red has a few drawbacks: First, phenol red is able to simulate the performance of a number of steroid hormones, primarily estrogen [14]. Therefore, when studying estrogen-sensitive cells like breast tissue, a medium free of phenol red is recommended. Sodium-potassium balance is disrupted by the presence of phenol red in several serum-free formulations. Adding serum or bovine pituitary hormone to the media may counteract this effect [15]. Thirdly, detection in flow cytometric experiments is hindered by the presence of phenol red.
Inorganic salts
Media containing inorganic salts, such as sodium, potassium, and calcium ions, assist maintain osmotic equilibrium and regulate membrane potential.
Amino acids
Since amino acids are the fundamental components of protein, they are an essential component of every single cell growth medium that has ever been conceived. Because cells are unable to produce certain amino acids on their own, it is important that the culture medium include essential amino acids. They are necessary for the proliferation of cells, and the concentration at which they are present determines the maximum cell density that may be attained. In particular, L-glutamine, an essential amino acid is especially crucial.
L-glutamine functions as a secondary source of energy for the metabolism and contributes nitrogen to the production of NAD, NADPH, and nucleotides. Due to the fact that L-glutamine is an unstable amino acid that, with time, changes into a form that cells are unable to utilize, it must be given to the medium.
In addition, non-essential amino acids may be supplied to the medium in order to refuel those that have been used up throughout the growth process. The growth of the cells is boosted and their viability is increased when the growth medium is supplemented with nonessential amino acids.
Carbohydrates
Carbohydrates in the form of sugars are the principal source of energy. Many of the media also include maltose and fructose in addition to the more common sugars of glucose and galactose.
Proteins and peptides
Albumin, transferrin, and fibronectin are the most commonly used proteins and peptides. They are especially significant in media that do not include serum. Albumin, transferrin, aprotinin, fetuin, and fibronectin are some of the proteins that may be found in serum, which is a rich supply of protein.
Albumin is the primary protein found in blood, and its function is to bind and transport various substances, including water, salts, free fatty acids, hormones, and vitamins, between different organs and cells. Albumin's ability to attach to chemicals makes it an effective candidate for removing harmful compounds from the medium in which cells are cultured.
Aprotinin is a protective agent in cell culture systems, since it is stable at neutral and acidic pH, as well as resistant to high temperatures and the destruction that may be caused by proteolytic enzymes. It is capable of inhibiting a number of serine proteases, including trypsin, amongst others.
Fetuin is a glycoprotein that may be detected in higher amounts in the serum of fetal and newborn animals compared to adult serum. In addition to that, it acts as a serine protease inhibitor. The protein fibronectin is an essential component in the process of cell adhesion. Transferrin is a protein that transports iron and is responsible for delivering iron to the membranes of cells.
Fatty acids and lipids
They play a crucial role in serum-free medium when serum is absent.
Vitamins
Numerous vitamins are necessary for cell development and proliferation. Vitamins cannot be produced in adequate amounts by cells and are thus essential in tissue culture as dietary supplements.
In cell culture, the serum is the primary source of vitamins; however, media are also treated with various vitamins to make them suited for a specific cell type. Most typically, the B group vitamins are used for growth stimulation.
Trace elements
Chemical elements such as copper, zinc, selenium, and tricarboxylic acid intermediates are known as trace elements. Trace elements are often added to media that does not include serum in order to replace those that are typically present in serum. These elements are important chemical components that are required for a healthy cell development. Many biochemical reactions depend on certain micronutrients, such as enzyme activity.
Medium supplements
The full growth medium suggested for certain cell lines needs extra components that are absent from the baseline media and serum. These dietary supplements support cell growth and appropriate metabolic function.
Although hormones, growth factors, and signaling molecules are essential for the appropriate proliferation of particular cell lines, the following precautions should always be taken: Since the addition of supplements might alter the osmolality of the complete growth medium, which can inhibit cell development, it is always advisable to verify the osmolality after adding supplements. For the majority of cell lines, the optimum osmolality ranges between 260 and 320 mOSM/kg.
Antibiotics
Antibiotics are often employed to inhibit the development of bacterial and fungal pollutants [16], although they are not essential for cell growth. Since antibiotics might conceal contamination by mycoplasma and resistant bacteria, their routine use is not suggested for cell culture [17, 18].
In addition, antibiotics may disrupt the metabolism of hypersensitive cells. The penicillin-streptomycin combinations made by MilliporeSigma and Life Technologies are often used. Plasmocin has been utilized in the culture of the glioma cell lines TS603, TS516, and BT260 [19], and it has been shown to be effective in removing mycoplasma contamination (20).
Serum
Albumins, growth factors, and growth inhibitors are all present in serum. Serum is one of the most significant components of cell culture medium because it provides amino acids, proteins, vitamins (especially fat-soluble vitamins such as A, D, E, and K), carbohydrates, lipids, hormones, growth factors, minerals, and trace elements.
Serum from fetal and calf bovine sources is often utilized to promote the development of cultured cells. Fetal serum is an abundant supply of growth factors and is suitable for cell cloning and the development of sensitive cells. Due to its diminished growth-promoting capabilities, calf serum is employed in contact-inhibition experiments. Normal growth mediums often include 2% to 10% serum. The addition of serum to culture medium serves the following purposes [21]:
-
The serum delivers the essential nutrients for cells (both in solution and attached to proteins).
-
Several growth factors and hormones involved in growth promotion and specialized cell activity are included in serum.
-
It offers many binding proteins, like albumin and transferrin, that transport other chemicals into the cell. For example, albumin delivers fats, vitamins, hormones, etc. into cells.
-
It also provides proteins, such as fibronectin, which increase cell adhesion to the substrate. Additionally, it produces spreading elements that aid in cell expansion before division.
-
It delivers protease inhibitors that prevent proteolysis in cells.
-
It also contains minerals such as Na+, K+, Zn2+, and Fe2+.
-
It enhances the viscosity of the media, so protecting the cells from mechanical injury during suspension culture agitation.
-
It's also a buffer.
References
[1] Morgan J, Morton H, Parker R. Nutrition of animal cells in tissue culture; initial studies on a synthetic medium. Proc Soc Exp Biol Med. 1950;73:1-8
[2] Kerbel R, Blakeslee D. Rapid adsorption of a foetal calf serum component by mammalian cells in culture. A potential source of artifacts in studies of antisera to cell-specific antigens. Immunology. 1976;31:881-91
[3] Sula K, Draber P, Nouza K. Addition of serum to the medium used for preparation of cell suspensions as a possible source of artifacts in cell-mediated reactions studied by means of the popliteal lymph node test. J Immunogenet. 1980;7:483-9
[4] Mariani E, Mariani A, Monaco M, Lalli E, Vitale M, Facchini A. Commercial serum-free media: hybridoma growth and monoclonal antibody production. J Immunol Methods. 1991;145:175-83
[5] Barnes D, Sato G. Methods for growth of cultured cells in serum-free medium. Anal Biochem. 1980;102:255-70
[6] Yu H, Lu S, Gasior K, Singh D, Vazquez Sanchez S, Tapia O, et al. HSP70 chaperones RNA-free TDP-43 into anisotropic intranuclear liquid spherical shells. Science. 2021;371:
[7] Meharena H, Marco A, Dileep V, Lockshin E, Akatsu G, Mullahoo J, et al. Down-syndrome-induced senescence disrupts the nuclear architecture of neural progenitors. Cell Stem Cell. 2022;29:116-130.e7
[8] Iscove N, Melchers F. Complete replacement of serum by albumin, transferrin, and soybean lipid in cultures of lipopolysaccharide-reactive B lymphocytes. J Exp Med. 1978;147:923-33
[9] Stoll T, Muhlethaler K, von Stockar U, Marison I. Systematic improvement of a chemically-defined protein-free medium for hybridoma growth and monoclonal antibody production. J Biotechnol. 1996;45:111-23
[10] Darfler F. A protein-free medium for the growth of hybridomas and other cells of the immune system. In Vitro Cell Dev Biol. 1990;26:769-78
[11] Barnes D, Sato G. Serum-free cell culture: a unifying approach. Cell. 1980;22:649-55
[12] Hamilton W, Ham R. Clonal growth of chinese hamster cell lines in protein-free media. In Vitro. 1977;13:537-47
[13] Zigler J, Lepe Zuniga J, Vistica B, Gery I. Analysis of the cytotoxic effects of light-exposed HEPES-containing culture medium. In Vitro Cell Dev Biol. 1985;21:282-7
[14] Berthois Y, Katzenellenbogen J, Katzenellenbogen B. Phenol red in tissue culture media is a weak estrogen: implications concerning the study of estrogen-responsive cells in culture. Proc Natl Acad Sci U S A. 1986;83:2496-500
[15] Karmiol S. Development of serum free media. In: Master JRW, editor. Animal Cell culture, 3rd ed. Oxford:Oxford University Press; 2000.
[16] Perlman D. Use of antibiotics in cell culture media. Methods Enzymol. 1979;58:110-6
[17] McGarrity G. Spread and control of mycoplasmal infection of cell cultures. In Vitro. 1976;12:643-8
[18] Masters J, Stacey G. Changing medium and passaging cell lines. Nat Protoc. 2007;2:2276-84
[19] Chakraborty A, Laukka T, Myllykoski M, Ringel A, Booker M, Tolstorukov M, et al. Histone demethylase KDM6A directly senses oxygen to control chromatin and cell fate. Science. 2019;363:1217-1222
[20] Molla Kazemiha V, Azari S, Amanzadeh A, Bonakdar S, Shojaei Moghadam M, Habibi Anbouhi M, et al. Efficiency of Plasmocin™ on various mammalian cell lines infected by mollicutes in comparison with commonly used antibiotics in cell culture: a local experience. Cytotechnology. 2011;63:609-20
[21] Kragh Hansen U. Molecular aspects of ligand binding to serum albumin. Pharmacol Rev. 1981;33:17-53